Blog - Tests and diagnosis
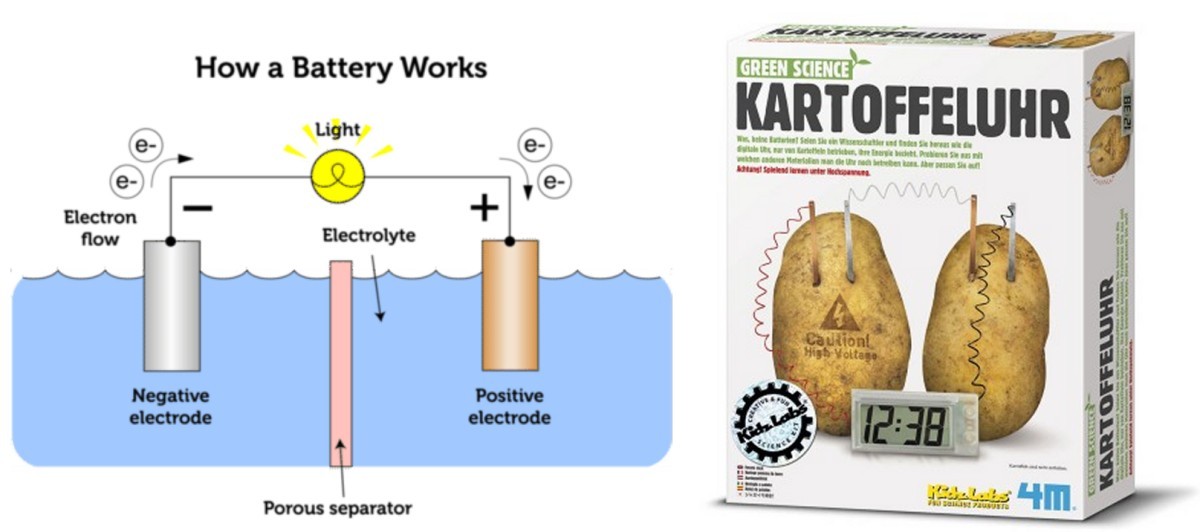
We are moving electrons. A lot of them!
When cells are charged and discharged, the volume of the mass inside the cell is changing. This is because the electrons have a mass and this mass is added or removed when charged or discharged.
How many electrons are moved when working with the current of 1 Amp?
The electron flow produces current. This is measured in amperes (A), although strictly speaking, electron flow produces a negative current because of the negative charge on it.
1 A = 1 C/s (coulomb per sec)
One unit of charge = 1.6 E-19 C
Hence, 1 Coulomb contains 1/(1.6 E-19)=6.25 E18 unit charges. That’s 6,250,000,000,000,000,000 units of charge. That’s how many electrons will flow in (the opposite direction of) a 1A current within 1 sec.
Imagine you discharge 100Ah cell for 1 hour with 100Amp. The total amount of electrons will be:
100 (Amp) x 3600 (s) x 6.25 E18 = 2,25 E24 of electrons.
Enjoy the life!
Note: there is a much more significant change of the internal structure of the cells due to the chemical process on the electrodes of the cells. That is why, simply speaking the cells work like muscles that stretch and expand when charged or discharged.

 English
English Česká republika
Česká republika Germany
Germany France
France España
España Italia
Italia Sverige
Sverige Polski
Polski Nederland
Nederland